AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Nucleus atom11/3/2022  
Commonly referred to as the ‘plum pudding model’.He described an atom as a positively shaped sphere with negatively charged electrons embedded into it.The claim that atoms are indivisible was later scrapped when subatomic particles were discovered.The structure of the atom was not appropriately explained.This theory fails to explain the existence of isotopes.Atoms can neither be created nor be destroyed.Atoms rearrange themselves during a chemical reaction.Each atom bears a constant mass that differs from element to element.Particular elements have only particular types of atoms in them.All matter is composed of atoms that are indivisible.These atoms and nuclei class 12 notes delve into their theories about the structure of the atom. Thomson, Ernest Rutherford, and Niels Bohr made significant contributions to the discipline. Each of these models had its own set of advantages and disadvantages, and they were all important in the creation of the present atomic model. Many scientists attempted to describe the structure of the atom using atomic models in the 18th and 19th centuries. This is why various elements have distinct qualities. As a result, their atomic structures differ. The total number of protons in an element’s nucleus is described by its atomic number.ĭifferent elements have different numbers of protons and electrons. The nucleus of the atom is made up of protons and neutrons, which are surrounded by the atom’s electrons. The structure of an atom refers to the constitution of its nucleus and the way electrons are arranged around the nucleus. In this ‘Atoms and nuclei study material’, we will be covering everything that you need to know about atoms and nuclei in detail, from their structure to the composition. Whereas, Nuclei are the central region of an atom that contains positively-charged protons and neutral neutrons which occupy most of the atom’s volume, except for a small space between the nucleus and an electron cloud called the orbital zone or nucleus. Atoms are defined as matter composed of subatomic particles such as protons, neutrons, and electrons. 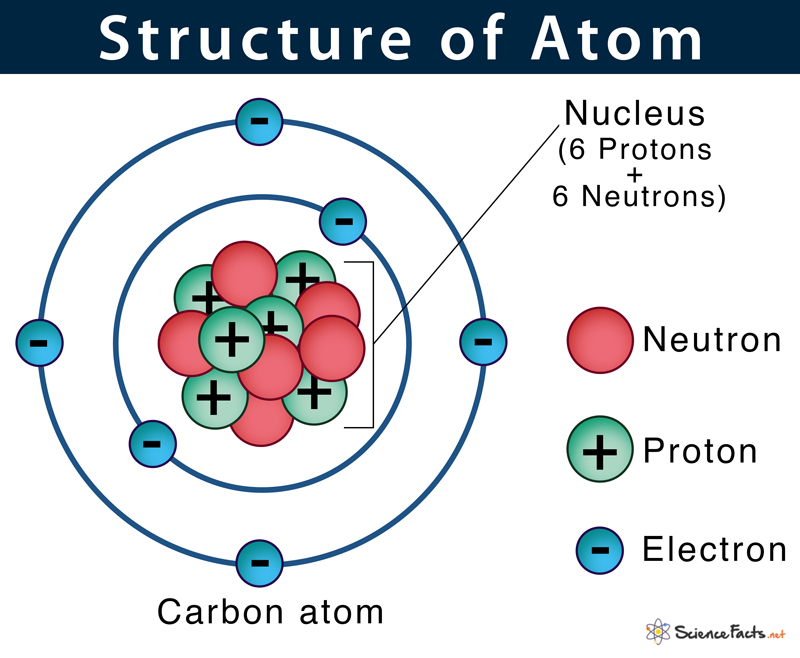
Atoms and nuclei are the fundamental building blocks of matter. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
